Aluminum:
Aluminum capacitors are the workhorses of big capacitors, they are small and cheap for their capacity and can be found
in sizes from <1 uF to over 1 farad. They are commonly available to 450 volts working voltage, with a few to at least
600 volts, much higher than other types of electrolytic capacitors. Aluminum electrolytic capacitors use a layer of
aluminum oxide (Al2O3 with a K of about 8.5) grown on aluminum foil. The aluminum foil forms one electrode while the
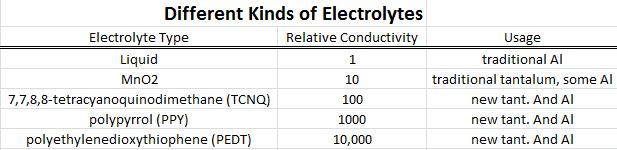
other is a non-aqueous electrolyte (hence the name) in a thin paper separator, and another foil layer for the cathode. The
separator holds the electrolyte and keeps the cathode from rubbing on the dielectric and damaging it. The classic
electrolyte formulas date back to the 1930s and were usually a glycol or amine (an organic solvent), in which a
conductive salt (a solute) is dissolved, plus 1-2% water. The water raises the electrolyte conductance and promotes
healing of the aluminum oxide. Many variations on this have been used over the years, although ethylene glycol is still
used. A chemical called gamma-butyrolactone is commonly used in high-temperature electrolytics. The various solutes
include benzoic acid, salicylic acid and adipic acid. A patent search will find any number of exotic electrolytes, but who
knows how many of these are actually in production. Given the variations that aluminum electrolytics are found in
however, there may be many propriety electrolytes in use. The electrolyte is specialized enough that many small pacific-
rim cap makers buy their electrolyte (often from Japan) rather than make it themselves. Some chemicals in the
electrolytes may be toxic to some extent. To make a non-polarized capacitor, the other foil electrode is also oxide
coated.
The reason for the electrolyte is that the surface of the oxide-coated foil electrode is etched to greatly increase the surface
area. The ratio of capacitance with etched foil vs unetched foil is called "foil gain", and is typically 10-200 for modern
aluminum electrolytics, depending on voltage. The other foil electrode can't make intimate contact with the dielectric by
itself so the electrolyte is used as an intermediary conductor. Etching the foil is a science in its own right. High-voltage
electrolytics require a different surface than low-voltage due to the different thickness in oxide coating required. And
either AC or DC may be used depending on the surface desired. There has been a slow-but-steady improvement in
etching that has improved volumetric efficiency over the years, roughly doubling over the last decade. As manufacturers
phase in the improved foil, they will obsolete the capacitor series that use the old foil and replace them with smaller parts
that use the new foil. However, the reduced-volume parts tend to have slightly higher ESRs than the equivalent older
higher-volume parts.
Almost all aluminum electrolytic capacitors are sealed with a crimped rubber plug (or "bung"). The common rubbers are
variations on ethylene propylene rubber (EPR), isobutylene-isoprene copolymer (butyl rubber), styrene butadiene rubber
(SBR), natural rubber (polyisoprene, NR), and ethylene propylene diene monomer (EPDM). EPDM is becoming more
popular, it has superior resistance to almost everything. Fillers are added to improve mechanical properties. Sealing the
capacitor has to be done just right for longest life. Too loose a seal can allow the capacitor to dry out. However,
aluminum electrolytics always generate some small amount of hydrogen and oxygen that must be vented off by diffusion
through the bung to prevent pressure buildup. And yet a very few hermetic electrolytics exist, but I don't know how they
do it.
Common cleaning solvents can be a problem for the plug and even for the aluminum can. Alcohols and water based
cleaners are generally OK but halogenated solvents will cause corrosion to the aluminum and none of the rubber
formulations will keep these solvents out. Freons, alkali cleaners, and strong petroleum solvents are also not
recommended. Methyl bromide is a special problem. A gas used for fumigation, methyl bromide is often used at ports
of entry and will penetrate common packaging including cardboard boxes and plastic bags, as well as the rubber bungs.
Some food processing plants like flour mills often use methyl bromide. Some electrolytics are made solvent-proof with
epoxy. Some manufacturers also offer a "solvent tolerant end seal" for short-term exposure to chlorinated solvents.
Manufacturers also warn against hydrogen sulfide, ammonia and various other chemicals.
Aluminum electrolytics have their problems, however; noise, high leakage, high temperature drift, high dielectric
absorption, high inductance, high almost everything bad. Low temperature is a problem for most aluminum capacitors.
For most types, capacitance falls off rapidly below room temperature while dissipation factor can be ten times higher at -
25C than at 25C.
Most limitations can be traced to the electrolyte. At high temperature, the water can be lost to evaporation, and the
capacitor (especially the small sizes) may leak outright. At low temperatures, the conductance of the salts declines,
raising the ESR, and the increase in the electrolyte´s surface tension can cause reduced contact with the dielectric. The
conductance of electrolytes generally has a very high temperature coefficient, +2%/C is typical, depending on size. The
electrolyte is implicated in various reliability issues as well.
Because of aluminum capacitor's problems, other capacitor types compete for aluminum's turf in some applications. In
small sizes, tantalum capacitors are available, but at a higher cost. Ceramic capacitors are available into the 100s of uF
and film capacitors are now available >1000 uF.
Aluminum capacitors are at their best doing simple jobs like line-frequency power supply filtering. If you want to use
them for more demanding applications, like switcher power supply filtering (low ESR), audio DC blocking (low-noise),
or in high temperature environments (long life), you must use care in selecting one the of special-purpose aluminum
capacitors. There are many to choose from, however; apparently the basic technology gives capacitor designers a lot of
room to maneuver. They include:
Low noise for audio applications.
Low leakage for RAM backup and even timing applications. Vendors sometimes tout these as tantalum
replacements.
Low equivalent series resistance (ESR) and/or low inductance (ESL) for switcher power supply filtering or for
high ripple-current applications. There are even 4-lead types available.
High-temperature types for improved life and reliability at elevated temperatures. Rated operating
temperatures go to 130C for common capacitors, but at the cost of increased size. 150C types for automotive
applications have started to appear. These have a special construction to resist vibration. Special types (rare)
have been available for operation to 200C.
Low-temperature types, as low as -65C. Very high and very low temperature rating may not be found in the
same capacitor.
Non-polarized for speaker crossover networks and other audio applications.
A variety of combination types such as high temperature + high reliability.
Photo-flash capacitors to handle very high surge currents that will destroy other electrolytics.
Miniature sizes and special shapes (tall and skinny, short and fat).
Axial leads, radial leads, screw terminals, tabs with holes for screws, quick-connect terminals and snap-in. DC
link aluminums will have multiple tabs attached to the foil to reduce the ESR and ESL.
SMD versions are now widely used in consumer electronics. Most SMD aluminum capacitors are just a
modified version of the conventional aluminum can. True SMD aluminums are stating to appear however. A
very few come in molded packages that might at first glance be mistaken for SMD tantalums.
Hermetic with very long life for military applications. See Cornell Dubilier. How they do it? The electrolyte is
not stated.